Over the past quarter century, we have come to realize that there is more to life than material goods and services, that "some of the best things in life are free." The pleasure we derive from breathing fresh air, drinking pure water, and enjoying the beauty that nature has provided is priceless and must not be sacrificed. Moreover, losing them will lead directly or indirectly to incalculable economic losses. We have come to appreciate the importance of our environment.
Much has been said and written about environmental problems with nuclear power, and they will be discussed at great length in this book. But in this chapter, we consider the wide variety of environmental problems in burning fossil fuels — coal, oil, and gas. They probably exceed those of any other human activity. The ones that have received the most publicity in recent years have been the "greenhouse effect," which is changing the Earth's climate; acid rain, which is destroying forests and killing fish; and air pollution, which is killing tens of thousands of American citizens every year, while making tens of millions ill and degrading our quality of life in other ways. We will discuss each of these in turn, and then summarize some of the other problems that have drawn lesser attention. But first we must begin with some basics.
Coal, oil, and gas consist largely of carbon and hydrogen. The process that we call "burning" actually is chemical reactions with oxygen in the air. For the most part, the carbon combines with oxygen to form carbon dioxide (CO2), and the hydrogen combines with oxygen to form water vapor (H20). In both of these chemical reactions a substantial amount of energy is released as heat. Since heat is what is needed to instigate these chemical reactions, we have a chain reaction: reactions cause heat, which causes reactions, which cause heat, and so on. Once started the process continues until nearly all of the fuel has gone through the process (i.e., burned), or until something is done to stop it. Of course, the reason for arranging all this is to derive the heat.
The carbon dioxide that is released is the cause of the greenhouse effect we will be discussing. A large coal-burning plant annually burns 3 million tons of coal to produce 11 million tons of carbon dioxide. The water vapor release presents no problems, since the amount in the atmosphere is determined by evaporation from the oceans — if more is produced by burning, that much less will be evaporated from the seas.
THE GREENHOUSE EFFECT
Electromagnetic radiation is an exceedingly important physical phenomenon that takes various forms depending on its wavelength. (The concept of wavelength is most easily understood for water waves, where it is the distance between successive crests. For electromagnetic waves it is the distance between successive peaks of the electric and magnetic fields.) Ordinary radios use the longest wavelengths of interest, 200 to 600 meters for AM stations. FM radio and television use wavelengths from a few meters (UHF) to less than 1 meter (VHF). Microwaves, familiar from the ovens that employ them, and radar, which plays a vital role in military applications and is also used by the police to catch speeders, have shorter wavelengths, in the range of centimeters to millimeters. Visible light is electromagnetic waves with much shorter wave lengths, ranging from 0.0004 millimeters for purple to 0.00055 millimeters for green to 0.0007 millimeters for red. Wavelengths between where visibility ends (0.0008 millimeters) and the microwave region begins (0.1 millimeters) are called infrared.
Every object in the universe constantly emits electromagnetic radiation, and absorbs (or reflects) that which impinges on it. According to the laws of physics, the wavelength of the emitted radiation decreases inversely as the temperature increases. (Here we use absolute temperature, which is Fahrenheit temperature plus 460 degrees). For example, the filament of a light bulb, which is typically at 6,000 degrees absolute, emits visible (yellow) light, while our bodies, which are normally at 559 degrees absolute (460 + 98.6), emit radiation of about 11 times longer wavelength, which is in the infrared region and therefore not visible — that's why we don't glow in the dark. More applicable to our discussion is the surface of the sun, which is at 11,000 degrees and therefore emits visible light as we can plainly see, and the surface of the Earth which, at about 520 degrees, emits infrared, as we know from the fact that it is not visible on a dark night.
The rate at which an object emits radiation energy increases very rapidly with increasing temperature (doubling the absolute temperature increases the radiation 16-fold). That's why a hand placed near a light bulb is heated much more by radiation from the filament when it is hot (light on) than when it is cool (light off). Now let us consider a bare object out in space, such as our moon. It receives and absorbs radiation from the sun, which increases its temperature, and this increased temperature causes it to emit more radiation. Through this process it comes to an equilibrium temperature, where the amount of radiation it emits is just equal to the amount it receives from the sun. That determines the average temperature of the moon. If this were the whole story, our Earth would be 54 degrees cooler than it actually is, and nearly all land would be covered by ice.
The reason for the difference is that the Earth's atmosphere contains molecules that absorb infrared radiation. They do not absorb the visible radiation coming in from the sun, so the Earth gets its full share of that. But a fraction of the infrared emitted by the Earth is absorbed by these molecules which then reemit it, frequently back to the Earth. That is what provides the extra heating. This is also the process that warms the plants in a greenhouse — the glass roof does not absorb the visible light coming in from the sun, but the infrared radiation emitted from the plants is absorbed by the glass and much of it is radiated back to the plants. That is how the process got its name — greenhouse effect. It is also the cause of automobiles getting hot when parked in the sun; the incoming visible radiation passes through the glass windows, while the infrared emitted from the car's interior is absorbed by the glass and much of it is emitted back into the interior.
Molecules in the atmosphere that absorb infrared and thereby increase the Earth's temperature are called greenhouse gases. Carbon dioxide is an efficient greenhouse gas. The atmosphere of Venus contains vast quantities of carbon dioxide, elevating its temperature by 500 degrees over what it would be without an atmosphere. That's why no astronaut will ever be able to land on Venus. Mars, on the other hand, has no atmosphere and is at the temperature expected from the simple considerations discussed above in connection with our moon.
Our problem is that burning coal, oil, and gas produces carbon dioxide, which adds to the supply already in the atmosphere, increasing the greenhouse effect and thereby increasing the temperature of the Earth. Prior to the industrial age, the concentration of carbon dioxide in the atmosphere was less than 280 ppm (parts per million). This was determined from analyzing air bubbles, trapped hundreds or thousands of years ago in the Antarctic and Greenland ice caps. By 1958 the carbon dioxide concentration had risen to 315 ppm, and by 1986 it was 350 ppm. The average temperature of the Earth has been about 1 degree warmer in the 20th century than in the 19th century, which is close to what is expected from this carbon dioxide increase. As the rate of burning coal, oil, and gas escalates, so too does the rate of increase of carbon dioxide in the atmosphere.
Predicting the increase of temperature expected from this is very complicated, but since it is so important, a great deal of effort has gone into deriving estimates. Results are usually discussed in terms of doubling the concentration of carbon dioxide, from 350 ppm to 700 ppm. If current trends continue, this will occur during the next century, perhaps as early as 2030.1 The direct effect of doubling the carbon dioxide in the atmosphere would be to raise the Earth's average temperature by 2.2°F. Two side effects will accentuate this temperature rise. One is that the increased temperature causes more water to evaporate from the oceans, which adds to the number of water molecules in the atmosphere; water vapor is also a greenhouse gas. The other is that there would be less ice and snow; these reflect away the visible light from the sun that would otherwise be absorbed by the Earth's surface.
There are many other factors of lesser importance that must be considered. Some of these factors tend to reduce the warming effect:
Yet other complicating processes accentuate the warming:
When all of these factors are taken into account as accurately as possible with our present knowledge, the best estimates are that doubling the carbon dioxide in the atmosphere will increase the average temperature by about 7°F. The uncertainty in this estimate is large; the true increase might be as little as 3 or as much as 15°F.
The importance of this greenhouse effect has become a public issue because of the recent abnormally hot summers accompanied by droughts that have severely reduced our agricultural output. Averaged over the Earth, the five warmest years in the past century have been in the 1980s, this despite the facts that the sun's energy output has been below normal and that there has been major volcanic activity, which would ordinarily reduce temperatures as explained above. Whether or not this recent abnormally warm weather is a manifestation of the increasing greenhouse effect is somewhat debatable, but unquestionably the greenhouse effect will become important sooner or later if we continue to use fossil fuels. There is, therefore, a strong consensus in both the scientific and environmentalist communities that the greenhouse effect should be given high-priority attention.
Since our emphasis here is the generation of electricity, we have concentrated our discussion on carbon dioxide from burning fossil fuels, but that is only part of the story on the greenhouse effect. Other aspects are discussed in the Chapter 3 Appendix. We now turn our attention to the consequences of this global warming.
PREDICTED CONSEQUENCES OF THE GREENHOUSE EFFECT
In December 1987, the U.S. Congress requested a report from the Environmental Protection Agency (EPA) on the health and environmental consequences expected from the greenhouse effect. The following discussion is based on that report.2
Agriculture is especially sensitive to climate. For example, the hot, dry summer of 1988 reduced corn yields in the Midwest by 40%. But with long-term planning, crops can be changed to compensate for climate change. Moreover, increased levels of carbon dioxide would have beneficial effects on agriculture, since carbon dioxide in the air is the principal source of material from which plants produce food.
The EPA's assessment is that the South will be hard hit, as the temperature becomes too hot for most of its crops, especially soybeans and corn. Florida is an exception, as citrus growing will be helped and tropical fruits can become a major new product. The Great Lakes region will be helped by the longer growing season. Crop yields in Minnesota will be increased 50-100%, with much lesser benefits elsewhere. Corn growing will become difficult in Illinois, but it can be replaced by sorghum. The Great Plains region will suffer the most, as it is already somewhat marginal for agriculture. Its major crops, wheat and corn, will probably have to be abandoned.
Live stock problems will increase. Heat stress will reduce breeding. Some of the live stock diseases that now plague the South will shift northward, and new tropical diseases will invade the South. Problems with agricultural pests will multiply. More pests will survive over the warmer winters, and they will breed more generations over the longer summers.
In summary, while there will be lots of disruptions and requirements for adjustment, the EPA does not expect food shortages to become critical in the United States. Problems could be more difficult in other parts of the world.
Forests will undergo some hard times. Each type of tree requires a specific climate. Thus, the growing area for each species will shift northward by 100 to 600 miles. This sounds innocuous, but adjustment periods will be difficult, with lots of die-off in the South and slow build-up in the North. Forests are constantly under stress from insects, diseases, competition with other plants, fires, wind, and the like. The added stress of changing climate is certain to cause trouble. A great deal of research and planning will be needed to cope with it.
A substantial fraction of U.S. cities are on seacoasts and thus close to sea level — Boston, New York, Baltimore, Washington, Miami, New Orleans, Houston, San Diego, Los Angeles, San Francisco, and Seattle, to name a few. If all snow and ice were to melt, sea level would rise by 270 feet, enough to inundate nearly all of these cities and vast other areas of the nation. If present trends continue, there will be a 20-foot rise in 200-500 years. A reasonable estimate for the middle of the next century is 1.5-3 feet. A 3-foot rise would flood major areas in Boston, New York, Charleston, Miami, and especially New Orleans, and in all would reduce the land area of the United States by an area equal to that of the state of Massachusetts. Most of this land loss would be in Louisiana and Florida. The barrier islands off our coasts, including Atlantic City and Miami Beach, would be in severe trouble.
In general, each 1-foot rise in sea level moves the coastline back 50 to 100 feet in the northeast, 200 feet in the Carolinas, 200 to 400 feet in California, 100 to 1,000 feet in Florida, and a few miles in Louisiana. The flooding now caused by storms once in 100 years would be expected about every 15 years. Since hurricanes are generated in warm water (above 79°F), more of them would be expected.
Inland penetration of salt water would cause lots of difficulties for aquatic life. This is already reducing oyster harvests in Chesapeake Bay. New York City derives its drinking water from the Hudson River just above the present penetration of salt water; that would have to be changed. Philadelphia and several other cities have similar problems. Contamination of groundwater with salt would be a widespread problem, especially in Florida.
For valuable land, like cities, effective measures can be taken to control flooding from rising sea levels. Dikes can be built with large capacity pump-out systems to handle overflow as in Holland. This can be used to protect major U. S. cities against a 6-foot rise in sea level for $30-100 billion. New buildings can be constructed on higher ground, back from the shore. Fill can be added to raise the land used for new construction. Again there will be lots of problems, but with wise planning, they can be solved or at least delayed.
There is also a concern for wetlands, which are important for waterfowl and some types of aquatic life. It would take only a small rise in sea level to cut the total U.S. wetlands in half.
Rising ocean levels do not necessarily mean rising levels in rivers and streams. For example, the levels of the Great Lakes are predicted to fall 2-5 feet, due to the greenhouse effect reducing rainfall and increasing evaporation. This will cause problems with shipping and with water supplies for cities and towns.
Wild animals and plants must adapt to climate changes, and there are many potential difficulties. In some situations, animals can simply move, but not always. The grizzly bears in Yellowstone park would have no place to go and probably would die out. Other casualties of the greenhouse effect will probably be panthers, bald eagles, and spotted owls.
Insect plagues can be expected to cause lots of problems for trees, as will increased floods and droughts. Forest fires will occur more frequently. Acid rain problems, to be discussed later, will become worse. It will not be an easy time for forests or the animals that inhabit them.
While all of these things are going on in the United States, the rest of the world will also be affected. When one thinks of rising sea levels, Holland comes to mind. Two-thirds of its land area is now below sea level, and even now it is protected by dikes against a 16-foot rise in sea level during storms. For $5-10 billion, Holland can be protected against a further 3-foot rise. Technology really helps in this matter. Bangladesh, with its low-technology society, is expected to suffer terribly from flooding as the sea level rises.
One would think that Canada would benefit from warmer climates, but there are many complications. With the oceans rising and the Great Lakes water levels falling, the St. Lawrence seaway will be in trouble. Southern Ontario, which has the most productive farmland in Canada, may suffer from drought as storm tracks and the rain they bring move north, and warmer temperatures cause increased evaporation. The western wheat belt will also be threatened by drought. But aside from these local problems, in general the greenhouse effect causes agriculture to move northward, and Canada can accommodate a lot of northward movement. The tree line moves north by about 35 miles for each degree Fahrenheit of global temperature rise.
Most of the above discussion is based on what will happen by the middle of the next century. But that is not the end of it. As long as we burn fossil fuels, the Earth's climate will continue to get warmer. The only solution is to strongly reduce our burning of coal, oil, and gas. Substituting nuclear energy for coal burning to generate electricity, and the substitution of electricity for oil and gas in heating buildings and to some extent in transportation, can play an important role in this process.
ACID RAIN
In addition to combining carbon and hydrogen from the fuel with oxygen from the air to produce carbon dioxide and water vapor, burning fossil fuels involves other processes. Coal and oil contain small amounts of sulfur, typically 0.5% to 3% by weight. In the combustion process, sulfur combines with oxygen in the air to produce sulfur dioxide, which is the most important contributor to acid rain. Air consists of a mixture of oxygen (20%) and nitrogen (79%), and at very high temperatures molecules of these can combine to produce nitrogen oxides, the other important cause of acid rain. Sulfur dioxide and nitrogen oxides undergo chemical reactions in the atmosphere to become sulfuric acid and nitric acid, respectively, dissolved in water droplets that eventually may fall to the ground as rain. This rain is therefore acidic.
Chemists measure acidity in terms of pH, on a scale which varies from 0 to 14. On this scale, a pH of 0 is maximum acidity, a pH of 7 is neutral, and a pH of 14 is maximum alkalinity, which is the opposite of acidity. Each unit of pH represents a factor of 10 in acidity. For example, a pH of 4 is 10 times more acidic than a pH of 5. Some examples of pH values are 1.2 for the sulfuric acid in automobile batteries, 2 for lemon juice or vinegar, 3 for apple juice, 4 for tomato juice, 5 for carrot juice, 6.3 for milk, 7.3 for blood, 8.5 for soap, and 13 for lye, which is almost a pure alkali.
We have seen that there is a substantial amount of carbon dioxide in the atmosphere. This dissolves in water droplets to form carbonic acid, familiar to us as carbonated water or soda water. As a result, "natural" rain is somewhat acidic, with a pH of about 5.6. Other natural factors, such as volcanic activity, also contribute, causing wide variations in pH. Rainfall pH as low as 4.0 has been observed even in places remote from the effects of fuel burning, like Antarctica and the Indian Ocean.
There is evidence that rain is made appreciably more acidic by the sulfuric and nitric acid from fossil fuel burning. For example, one study indicated that in the eastern United States, the pH of rain was in the range 4.1-4.5 in the 1980s versus 4.5-5.6 in the 1950s.
After the rain falls, it percolates through the ground, dissolving materials out of the soil. This alters its pH and introduces other materials into the water. If the soil is alkaline, the water's acidity will be neutralized, but if it is acid, the acidity of the water may increase. This water is used by plants and trees for their sustenance, and eventually flows into rivers and lakes. There have been various reports indicating that streams and lakes have been getting more acidic in recent years, although the effects seem to be highly variable and not closely correlated with releases of sulfur dioxide and nitrogen oxides. A study of Adirondacks lakes between 1975 and 1985 found that one-quarter became more acid, one-quarter (including some less than 5 miles away) became less acid, and the rest were unchanged. These findings were clearly inconclusive. Among other thins, acid rain most frequently discussed are makes lakes unlivable for fish and other aquatic life and destroys forests. On both of these matters, the evidence is highly complex and took many years to develop, and it is still not completely unequivocal. But by now most scientists are convinced that the effects are real and serious in some areas.
One of the problems is that it is difficult to be certain about any specific area. Of the 50,000 lakes in the northeastern United States, 220, mostly in the Adirondacks mountains of New York, have no fish because the water is too acidic. Since tourism is very important in the Adirondacks region, this has had serious economic consequences that may be ascribed to acid rain, and the residents are very upset about it. On the other hand, there is no evidence that there ever were fish in those lakes. The surrounding soil is naturally highly acidic, which is surely at least partially responsible for the lakes' acidity. Moreover, there is no indication that the acidity has been changing in recent years.
A National Academy of Sciences committee3 investigated the problem by estimating the acidity prior to 1800. They analyzed fossil remains of microorganisms in the lake bottom sediments. Of the nine Adirondacks lakes for which these data and information on fish populations are available, six showed clear signs of increased acidity. For example, the pH of Big Moose Lake fell from 5.8 prior to 1800 to 4.9 at present, and the number of fish species in the lake declined from 10 in 1948 to 5 or 6 in 1962. It would normally take many hundreds or thousands of years for this large a change to take place through natural processes. The National Academy of Sciences Committee concluded that acid rain is responsible for these changes.
The problem of forest destruction is at least equally complicated. The most elaborate study was done in Germany, where a serious blight on trees has been in action.4 In 1982 it affected 8% of all West German trees, and by 1987, 52% were affected. Since Germans revere their forests, this is a high-priority issue for them. At the time when the political issue of installation of Pershing nuclear missiles on German soil reached its highest pitch, a poll found that the death of forests was the greatest concern of the German public.
A study of one particular forest concluded that the trees were under stress from a variety of factors but acid rain was "the straw that broke the camel's back." It dissolved aluminum out of the soil — about 5% of all soil is aluminum — and this toxic element was picked up by the roots of trees. Not only did the toxicity of aluminum cause direct damage to the trees, but aluminum was picked up instead of calcium and magnesium, which are crucial to a tree's nutrition. The problem was compounded by the nitric acid in the rain acting as fertilizer to accelerate the trees' growth at a time when important nutrients were lacking. Because of this stress, the trees were succumbing to what would normally be non-lethal attacks by insects compounded by drought.
Trees are suffering blight in many parts of the world, and acid rain is suspected of contributing to the problem. A prime example are spruce trees in the Appalachian mountains, which are dying off from New England to North Carolina. In this situation, the trees are at a high altitude where they are engulfed in a mist a large fraction of the time. Acidity in the mist is believed to be an important contributor to the damage — acidity that has the same origin as acid rain, mainly emissions from coal-burning plants.
Some of the most important problems caused by acid rain are political. The sulfur dioxide and nitrogen oxides that cause acid rain originate far away, in other states or in other countries. The acid rain that is damaging lakes and forests in the eastern United States and Canada originates in coal-burning power plants in the Midwest. (Midwestern lakes and forests are not damaged by these emissions because soil in that region is naturally less acidic.) The acid rain that damages lakes and forests in Scandinavia originates in Britain and Western Europe, and the latter also contributes to damaging German forests. It is only natural for people to be very upset by losses caused by others, and they are demanding action.
Acid rain from U.S. sources is a top priority political issue in Canada. In any meetings between leaders of the two countries, the Canadians insist that it be high on the agenda. They insist on U.S. action to reduce sulfur dioxide emissions, and because of the high value we place on Canadian friendship, they will probably get it. In 1989, the Bush Administration introduced new clean air legislation that requires cutting sulfur dioxide emissions in half by the end of the century. It is estimated that this will increase the cost of electricity from coal burning by about 20%. This still does not face the problem of nitrogen oxides, for which there is no very effective control technology.
AIR POLLUTION
The greenhouse effect and acid rain have received more media attention and hence more public concern than general air pollution. This is difficult to understand, because the greenhouse effect causes only economic disruption, and acid rain kills only fish and trees, whereas air pollution kills people and causes human suffering through illness.
We have already described the processes that produce sulfur dioxide and nitrogen oxides, which are important components of air pollution as well as the cause of acid rain. But many other processes are also involved in burning fossil fuels. When carbon combines with oxygen, sometimes carbon monoxide, a dangerous gas, is produced instead of carbon dioxide. Thousands of other compounds of carbon, hydrogen, and oxygen, classified as hydrocarbons or volatile organic compounds, are also produced in the burning of fossil fuels. During combustion, some of the carbon remains unburned, and some other materials in coal and oil are not combustible; these come off as very small solid particles, called particulates, which are typically less than one ten thousandth of an inch in diameter, and float around in the air for many days. Smoke is a common term used for particulates large enough to be visible. Some of the organic compounds formed in the combustion process attach to these particulates, including some that are known to cause cancer. Coal contains trace amounts of nearly every element, including toxic metals like beryllium, arsenic, cadmium, selenium, and lead, and these are released in various forms as the coal burns.
All of the above pollutants are formed and released directly in the combustion process. Some time after their release, nitrogen oxides may combine with hydrocarbons in the presence of sunlight to form ozone, one of the most harmful pollutants. Or other compounds may form, such as PAN, which is best known as the cause of watering eyes in Los Angeles smog.
Let us summarize some of the known health effects of these pollutants:5
It is well recognized that toxic substances acting in combination can have much more serious effects than each acting separately, but little is known in detail about this matter. Information on the quantities of air pollutants required to cause various effects is also very limited. However, there can be little doubt that air pollution is a killer.
The clearest evidence linking air pollution to increased mortality comes from several catastrophic episodes6 in which a large number of excess deaths occurred during times of high pollution levels, in all cases caused by coal burning in association with unfortunate weather conditions. In a December 1930 episode in the Meuse Valley of Belgium, there were 60 excess deaths and 6,000 illnesses. In an October 1948 episode in Donora, Pennsylvania, there were 20 deaths (versus 2 normally expected) in a 4-day period during which 6,000 of the 14,000 people in the valley became ill. There were at least eight episodes in London between 1948 and 1962 in each of which hundreds of excess deaths were recorded, the largest in December 1952 when 3,500 died. There were three episodes in New York City involving over a hundred deaths, one in November 1953 causing 360 deaths, another in January-February 1963 leading to 500 deaths, and a third in November 1966 responsible for 160 deaths. In all of these cases, the mortality rates rose sharply when measured air pollution levels reached very high values, and fell when the latter declined.
 |
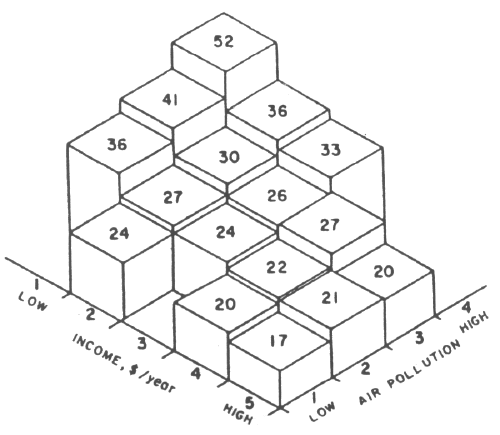
| Fig. 1 — Mortality rate (deaths/year/1000 population) for males aged 50-69 versus annual income and air polution in the section of Buffalo, New York in which they live. |
The best method for establishing a connection between "normal" levels of air pollution and premature mortality is through comparison of mortality rates between different geographic areas with different average air pollution levels. Of course there are other factors affecting mortality rates that vary with geographic area, like socioeconomic conditions; the data must be analyzed thoroughly to eliminate these factors. As an example, Fig. 1 shows a plot of annual mortality rates for males aged 50-69 in various census tracts of Buffalo, New York, during 1959-1961, versus average annual income and average air pollution.7 It is evident from Fig. 1 that for each income range, mortality rates increased with increasing pollution level. A mathematical analysis separates the two effects, giving the risk of air pollution alone. There have been a number of other such studies,8 comparing various cities in the United States, all the counties in the United States, various cities in England, and so on. In addition, there have been a number of studies9 of mortality rates in a given city, especially New York and London (also in several other American cities and Tokyo), on a day-by-day basis, correlating them with air pollution levels. In these there are no complications from socioeconomic factors, since these do not vary on a day-to-day basis. However, there are weather factors that must be removed by mathematical analysis in order to determine the effects of air pollution alone.
These studies have established strong correlations in timing between elevated air pollution levels and mortality rates. There are also numerous studies10 of temporary illness, involving hospital admissions, questionnaires, measurements of pulmonary function, and so on, in New York, London, Chicago, five Japanese cities, Rotterdam, Oslo, and others, all indicating strong correlations with abnormally high air pollution levels.
The U.S. Department of Energy's Office of Health and Environmental Research sponsored a multiyear study by a Harvard University research group to evaluate all of the available studies. Its conclusion was that air pollution is probably causing about 100,000 deaths per year in the United States.11 These deaths are principally from heart and lung disease. In addition it is estimated that air pollution causes about 1,000 cancer deaths per year.6
The estimate of 100,000 deaths per year means that 1 American out of 30 dies as a result of air pollution. Most environmental agents that get abundant media attention and public concern, such as Alar in apples, pesticides that have been banned, PCBs, and formaldehyde, give those exposed less than one chance in 100,000 of dying from their effects. We see that air pollution, which gives 1 chance in 30, is thousands of times more harmful.
While the evidence for health effects from air pollution is undeniable, reaching an understanding of them has proved to be a very difficult task. Historically, the pollutants most easily and therefore most frequently measured were sulfur dioxide and suspended particulate matter; thus, nearly all correlation studies were based on them. Until the late 1970s, it was widely assumed that these were the materials actually responsible for the health damage. Yet animals exposed to very high levels of these materials for long time periods showed no ill effects. Moreover, men occupationally exposed to 100 times the normal outdoor levels of sulfur dioxide from refrigeration sources, oil refineries, and pulp and paper mills were not seriously affected. Occupational settings where suspended particulate levels are 100 times higher than the outdoor average revealed no important health effects.10 In response to these findings, in the mid-1970s there was a strong trend toward designating sulfate particulates, resulting from chemical reactions of sulfur dioxide with other chemicals in air, as the culprit, and some still continue to support that viewpoint; but animals show no ill effects even from prolonged exposure to relatively high levels of sulfate particulates.
One could concentrate on other components of air pollution as possible sources of the health effects, and there are plenty of candidates to choose from. Most of them, including the great majority of volatile organic compounds, have not been investigated as causes of health effects. But there is no evidence or strong body of opinion that any one substance is the major culprit in air pollution. Health effects probably arise from complicated interactions of many pollutants acting together. We may never understand the process in any detail.
The greatest difficulty in trying to tie down causes is that air pollution doesn't kill healthy people in one fell swoop. It rather continuously weakens the respiratory and cardiovascular systems over many decades until they collapse under one added insult. This explains why there are no mortality effects on the occupationally exposed, on animals, and on college students used as volunteer subjects in controlled tests.10
Since we do not know what components of air pollution cause the health effects, it is impossible to know what pollution control technologies will be effective in averting them. The often-heard statement "we can clean up coal burning" involves a large measure of wishful thinking.
How much of this air pollution can be averted by use of nuclear power? None of the pollutants discussed above are released by nuclear reactors. Coal burning, which now generates most of our electricity, is by far the most polluting process. According to EPA estimates,5 fossil fuel burning in electric power plants produces 64% of all U.S. releases of sulfur dioxide, 27% of the particulates, and 31% of the nitrogen oxides, but less than 1% of the carbon monoxide or hydrocarbons. It is therefore reasonable to estimate that this is causing 30,000 of the 100,000 deaths per year caused by air pollution. Industrial fuel combustion, which is being rapidly replaced by electricity, produces 12% of the sulfur dioxide, 10% of the particulates, 22% of the nitrogen oxides, and 5% of the volatile organic compounds. Averting these emissions would make a very substantial contribution to the solution of our air pollution problems. If electric cars were to be successful and more electrically powered buses and railroads were used, further great gains would be achieved since transportation is responsible for 84% of the carbon monoxide, 41% of the volatile organic compounds, and 40% of the nitrogen oxides.
We are spending an estimated $30 billion per year to reduce air pollution, and a substantial fraction of it is due to coal-burning power plants. Replacing them with nuclear plants would therefore save many billions of dollars per year in this area alone.
OTHER ENVIRONMENTAL EFFECTS OF FOSSIL FUELS12
Sixty percent of our coal is now strip-mined. That is, huge earth-moving machines strip off the covering soil to reach the coal and then scoop it up and load it into trucks. In a single bucket load these machines pick up 300 tons. They sometimes remove close to 200 feet of covering soil to reach the coal. This is earth moving on a gigantic scale, and in the process, the land is badly scarred. Most states now have laws requiring that the land contour be restored and revegetated. These laws have done a lot to improve the situation, but the restoration often leaves much to be desired. Our largest coal reserves are in the Wyoming-Montana region, where restoration is particularly difficult because of sparse rainfall. Strip-mine operators are required to post a bond as evidence of intent to restore the land after mining, but it is said that no coal company has ever gotten its reclamation performance bond returned in Montana.12 Over a million acres of strip-mined land is awaiting reclamation, and new land is being strip-mined at a rate of 65,000 acres per year.
The remaining 40% of our coal comes from underground mines, and that percentage will eventually have to increase as locations for strip mining run out — the great majority of our coal reserves require underground mining. One of the environmental impacts of this endeavor is acid drainage from abandoned mines. Water seeping in reacts with sulfur compounds to produce sulfuric acid, which eventually seeps out and gets into streams, making them acidic. This kills fish and makes the water unfit for drinking, swimming, and many industrial applications. There are methods for preventing this acid mine drainage, but they are quite expensive and are not generally being implemented.
Another environmental impact of underground mining is land subsidence (ground on the surface moving downward as the abandoned mines below cave in), causing buildings on the surface to crack or even to be completely destroyed. Something like one-fourth of the 8 million acres that are above coal mines have subsided. About 7% of this subsidence has been in cities, where damage is very high and tragic to the homeowners. Subsidence in rural areas can change drainage patterns and make land unfit for farming. At the very least it scars the land. Laws governing mineral rights relieve mining companies of any responsibility for damage done by subsidence.
Another environmental impact of underground mining is fires that start by accident and are very difficult to put out. Some have smoldered for many decades. They release smoke laden with air pollutants, and their heat kills the vegetation. Of course, they also destroy a lot of coal. In 1983 there were 261 uncontrolled mine fires smoldering in the United States.
Coal is often washed just outside the mine to remove foreign materials, and the waste material from this washing is piled up in unsightly heaps. In 1983 there were 177,000 acres of these waste banks in the United States, the great majority in Appalachia. Many of these waste banks catch fire and burn, serving as a source of air pollution.
Mining coal is among the most unpleasant of occupations. A miner is in constant intimate contact with dirt, often without room to stand up, and engulfed in dust. Idealists have called it a job unfit for humans. Although there have been many improvements in recent years, it is still one of the most dangerous occupations, regularly killing over 100 men per year in the United States. (Early in this century it was killing well over 1,000 per year.)
But the most important health impact of coal mining is black lung disease, a name derived from the fact that, on autopsy, the lungs of coal miners are invariably found to be black. That disease, which causes lots of discomfort, it is not fatal. However, it leaves miners exceptionally susceptible to emphysema and a variety of other lung diseases.13 Underground miners have more than 20 times the normal risk of death from silicosis or pneumoconiosis, lung diseases caused by exposure to dust, and about 2.5 times the normal risk of dying from bronchitis, pneumonia, or tuberculosis. At younger ages, coal miners are healthier than the average person their age, but as they age the situation reverses; coal miners die an average of 3 years younger than the rest of the population of the same socioeconomic status.
The most publicized environmental effect of using petroleum as a fuel is oil spills, highlighted by the spill of 40,000 tons of oil from the tanker Exxon Valdez off the coast of Alaska in 1989. Although over a billion dollars was spent in the clean-up, many of the beaches were ruined and numerous species of aquatic animals suffered damage that will not be healed for decades. But by world standards this was not a large oil spill. In 1979, the Atlantic Empress was involved in a collision off the coast of Tobago in the Caribbean, spilling 305,000 tons, and in 1978 the Amoco Cadiz ruined many miles of French beaches with a spill of 237,000 tons. There are lots of smaller spills. U.S. tankers alone spilled an average of 215,000 tons per year in 1970-1974, and 380,000 tons per year in 1975-1979. At any given time, over 100 million tons of oil is being transported by ships, so it is not surprising that some of it occasionally ends up in the water.
Accidents on land can also spill oil into the oceans. The most spectacular case of this type was in Campeche Bay, Mexico, in 1979, where a well could not be capped for 280 days, during which it spilled 700,000 tons of oil into the Gulf of Mexico, doing heavy damage to aquatic life.
Our news media give much more publicity to spills off U.S. shores. Perhaps the most publicized was the 1969 spill off the California coast near Santa Barbara, in which 7,000 tons went into the water before the well was capped. The damage the oil did to the beautiful beaches there stopped off-shore drilling in that region for 20 years.
It seems clear that as long as we use oil heavily, we will pollute the oceans with it, and damage the local aquatic life. With the biggest spills to date off our shores being 7,000 tons in 1969 and 40,000 tons in 1989, we have perhaps been abnormally lucky.
CONCLUSIONS
In the last chapter, we showed that lots of new power plants will be needed in the United States in the near future, and that they will have to be nuclear or burners of fossil fuels. In this chapter we have reviewed some of the drawbacks to the latter, and we have seen that they are very substantial. These should be kept in mind as we consider the environmental problems with nuclear power in later chapters.
CHAPTER 3 APPENDIX
More about the Greenhouse Effect14
Carbon dioxide is responsible for only about half of the Earth's greenhouse effect warming. Methane, which is increased by escaping natural gas, contributes 18%, and nitrogen oxides, which are products of fossil fuel burning, account for 6%. Another major contributor is chlorinated fluorocarbons (CFCs), which are responsible for 14%. These are familiar as the working material in air conditioners, refrigerators, and freezers, and as the propellant in spray cans. (Other important sources of methane include flooded rice paddies and cows burping up gas from their stomachs.)
Another cause of the greenhouse effect is cutting down forests, since plants take carbon dioxide out of the air. Clearing of the Amazon rainforest is believed to be especially serious. Worldwide, an area of forests equal to the area of the state of Virginia is cleared every year. This is estimated to cause 20% of the greenhouse effect.
Burning wood (or biomass) releases carbon dioxide, but it does not contribute to the greenhouse effect because the wood was created from carbon dioxide, which the tree leaves absorb from the atmosphere.
In 1950, the United States was responsible for 45% of the 1.6 billion tons of carbon dioxide emitted throughout the world, but by 1980 world emissions increased to 5.1 billion tons with only 27% from the United States; Western Europe's share was 23% in 1950 and 16.5% in 1980. Increased use of fossil fuels in underdeveloped countries has played an important role in increasing emissions.